Role of hypoxia in obesity-induced disorders of glucose and lipid metabolism in adipose tissue - PubMed
- ️Invalid Date
Role of hypoxia in obesity-induced disorders of glucose and lipid metabolism in adipose tissue
Jun Yin et al. Am J Physiol Endocrinol Metab. 2009 Feb.
Abstract
Recent studies suggest that adipose tissue hypoxia (ATH) may contribute to endocrine dysfunction in adipose tissue of obese mice. In this study, we examined hypoxia's effects on metabolism in adipocytes. We determined the dynamic relationship of ATH and adiposity in ob/ob mice. The interstitial oxygen pressure (Po(2)) was monitored in the epididymal fat pads for ATH. During weight gain from 39.5 to 55.5 g, Po(2) declined from 34.8 to 20.1 mmHg, which are 40-60% lower than those in the lean mice. Insulin receptor-beta (IRbeta) and insulin receptor substrate-1 (IRS-1) were decreased in the adipose tissue of obese mice, and the alteration was observed in 3T3-L1 adipocytes after hypoxia (1% oxygen) treatment. Insulin-induced glucose uptake and Akt Ser(473) phosphorylation was blocked by hypoxia in the adipocytes. This effect of hypoxia exhibited cell type specificity, as it was not observed in L6 myotubes and betaTC6 cells. In response to hypoxia, free fatty acid (FFA) uptake was reduced and lipolysis was increased in 3T3-L1 adipocytes. The molecular mechanism of decreased fatty acid uptake may be related to inhibition of fatty acid transporters (FATP1 and CD36) and transcription factors (PPARgamma and C/EBPalpha) by hypoxia. The hypoxia-induced lipolysis was observed in vivo after femoral arterial clamp. Necrosis and apoptosis were induced by hypoxia in 3T3-L1 adipocytes. These data suggest that ATH may promote FFA release and inhibit glucose uptake in adipocytes by inhibition of the insulin-signaling pathway and induction of cell death.
Figures

Hypoxia in adipose tissue of ob/ob mice. A: P
o2 in epididymal fat pads of lean and ob/ob mice was determined with an oxygen meter equipped with a fiber optic oxygen sensor. The assay was conducted in mice at 6, 9, and 12 wk in age. Fat composition and body weight of the mice were determined (n = 3). B: hypoxia response gene expression in epididymal fat of ob/ob mice was determined in quantitative RT-PCR. The assay was conducted in mice at 6 and 12 wk in age (n = 3). C: protein levels in epididymal fat of lean and ob/ob mice were examined in a Western blot (n = 3). *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.001 vs. lean mice; #P < 0.05, ##P < 0.01 vs. 6-wk-old (6w) mice.
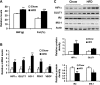
Hypoxia-induced gene expression in diet-induced obesity. A: after 8 wk on a high-fat diet (HFD), mice were examined for body weight (BW) and fat composition. B: hypoxia-induced gene expression in epididymal fat of lean and obese mice was determined using quantitative RT-PCR. C: protein levels in epididymal fat of HFD mice were examined by Western blot. *P < 0.05, **P < 0.01, ***P < 0.001 vs. chow diet-fed control mice.

Inhibition of insulin-induced glucose uptake by hypoxia. A: 3T3-L1 adipocytes were exposed to hypoxia (1% oxygen), and proteins were determined by Western blot in a time course study (n = 3 or 6). B: 2-deoxy-
d-[3H]glucose uptake. Uptake was examined in 3T3-L1 adipocytes after hypoxia treatment for different times. The assay was conducted in normoxic condition immediately after hypoxia treatment (n = 3). C: 15 min prior to cell collection, 100 nM insulin was added to normoxia- or hypoxia-treated 3T3-L1 adipocytes. Western blot was conducted to detect the phosphorylation of Akt in whole cell lysate. 0 represents normoxic condition. *P < 0.05, **P < 0.01, ***P < 0.001 vs. normoxic control (CON or 0 h).
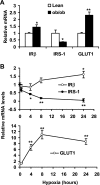
IRβ, IRS-1 and GLUT1 gene expression in adipose tissue and hypoxia-treated 3T3-L1 adipocytes. A: mRNA in epididymal fat pads of lean and ob/ob mice were determined with real-time quantitative RT-PCR. B: mRNA levels in 3T3-L1 adipocytes were determined with real-time quantitative RT-PCR after hypoxia treatment. *P < 0.05, **P < 0.01 vs. lean mice or untreated cells (0 h).
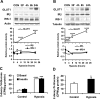
Hypoxia effects on L6 myotubes and βTC6 cells. A: effect of hypoxia on protein levels in L6 myotubes was investigated in a time course study in a Western blot (n = 3). B: hypoxia effect on protein levels in βTC6 cells (n = 3). C: 2-deoxy-
d-[3H]glucose uptake was performed in L6 myotubes after hypoxia exposure for 24 h (n = 3). D: glucose uptake was performed in βTC6 cells after hypoxia treatment for 24 h (n = 3). *P < 0.05, ***P < 0.001, ****P < 0.0001 vs. control (CON) cells or 0 h.
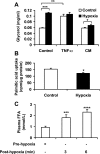
Hypoxia regulation of lipid metabolism. A: lipolysis was evaluated with glycerol release into culture medium of 3T3-L1 adipocytes. The assay was conducted after exposure to hypoxia or TNF-α (20 ng/ml) for 24 h (n = 4). Fresh 3T3-L1 adipocytes were cultured in conditioned medium (CM) made from supernatant of 3T3-L1 adipocytes treated by normoxia or hypoxia for 24 h. The net increase in glycerol level was obtained by comparing glycerol concentrations before and after 24-h culture in CM. B: [1-14C]palmitic acid uptake was performed in 3T3-L1 adipocytes after hypoxia treatment for 24 h (n = 3). C: FFA in femoral artery of lean rats. The femoral artery was clamped to block blood flow for 15 min to generate a hypoxia response in vivo. Blood samples were collected from the femoral vein before surgery and after clamp to determine the change in FFA (n = 11). *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001.

Hypoxia regulation of lipid metabolism-related proteins. A: protein levels in 3T3-L1 adipocytes were examined by Western blot after hypoxia treatment for 24 h (n = 6). B: protein levels in epididymal fat pad of ob/ob mice were examined by Western blot (n = 4 or 3). *P < 0.05, **P < 0.01, ***P < 0.001.
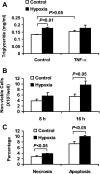
Hypoxia induction of adipocyte death. A: adipocyte death was examined indirectly with triglyceride release into CM of 3T3-L1 adipocytes exposed to hypoxia and/or TNF-α (20 ng/ml) for 24 h (n = 4). B: nonviable adipocyte number was examined with trypan blue staining after hypoxia treatment for 8 and 16 h (n = 4). C: the necrosis and apoptosis ratios in 3T3-L1 adipocytes were determined using flow cytometry after cells were treated with normoxia or hypoxia for 24 h.
Similar articles
-
Ye J, Gao Z, Yin J, He Q. Ye J, et al. Am J Physiol Endocrinol Metab. 2007 Oct;293(4):E1118-28. doi: 10.1152/ajpendo.00435.2007. Epub 2007 Jul 31. Am J Physiol Endocrinol Metab. 2007. PMID: 17666485
-
Dai S, Hong Y, Xu J, Lin Y, Si Q, Gu X. Dai S, et al. Biomed Pharmacother. 2018 Apr;100:93-100. doi: 10.1016/j.biopha.2018.01.111. Epub 2018 Feb 6. Biomed Pharmacother. 2018. PMID: 29425748
-
Tepaamorndech S, Kirschke CP, Pedersen TL, Keyes WR, Newman JW, Huang L. Tepaamorndech S, et al. FEBS J. 2016 Jan;283(2):378-94. doi: 10.1111/febs.13582. Epub 2015 Dec 12. FEBS J. 2016. PMID: 26524605
-
Adipose tissue oxygen tension: implications for chronic metabolic and inflammatory diseases.
Goossens GH, Blaak EE. Goossens GH, et al. Curr Opin Clin Nutr Metab Care. 2012 Nov;15(6):539-46. doi: 10.1097/MCO.0b013e328358fa87. Curr Opin Clin Nutr Metab Care. 2012. PMID: 23037900 Review.
-
Halleux CM, Servais I, Reul BA, Detry R, Brichard SM. Halleux CM, et al. J Clin Endocrinol Metab. 1998 Mar;83(3):902-10. doi: 10.1210/jcem.83.3.4644. J Clin Endocrinol Metab. 1998. PMID: 9506746 Review.
Cited by
-
Metabolic consequences of high-fat diet are attenuated by suppression of HIF-1α.
Shin MK, Drager LF, Yao Q, Bevans-Fonti S, Yoo DY, Jun JC, Aja S, Bhanot S, Polotsky VY. Shin MK, et al. PLoS One. 2012;7(10):e46562. doi: 10.1371/journal.pone.0046562. Epub 2012 Oct 1. PLoS One. 2012. PMID: 23049707 Free PMC article.
-
Huang W, Zhang J, Zou J, Wang X, Xu H, Guan J, Yi H, Liu S, Yin S. Huang W, et al. iScience. 2023 Jan 14;26(2):105985. doi: 10.1016/j.isci.2023.105985. eCollection 2023 Feb 17. iScience. 2023. PMID: 36798439 Free PMC article.
-
van der Kallen CJ, van Greevenbroek MM, Stehouwer CD, Schalkwijk CG. van der Kallen CJ, et al. Apoptosis. 2009 Dec;14(12):1424-34. doi: 10.1007/s10495-009-0400-4. Apoptosis. 2009. PMID: 19757063 Free PMC article. Review.
-
CYP1A1, VEGFA and Adipokine Responses of Human Adipocytes Co-exposed to PCB126 and Hypoxia.
Amine ZE, Mauger JF, Imbeault P. Amine ZE, et al. Cells. 2022 Jul 24;11(15):2282. doi: 10.3390/cells11152282. Cells. 2022. PMID: 35892579 Free PMC article.
-
Li G, Lu WH, Ai R, Yang JH, Chen F, Tang ZZ. Li G, et al. Cardiovasc Diabetol. 2014 Feb 24;13:52. doi: 10.1186/1475-2840-13-52. Cardiovasc Diabetol. 2014. PMID: 24564828 Free PMC article.
References
-
- Bano KA, Batool A. Metabolic syndrome, cardiovascular disease and type-2 diabetes. J Pak Med Assoc 57: 511–515, 2007. - PubMed
-
- Bashan N, Burdett E, Hundal HS, Klip A. Regulation of glucose transport and GLUT1 glucose transporter expression by O2 in muscle cells in culture. Am J Physiol Cell Physiol 262: C682–C690, 1992. - PubMed
-
- Bastard JP, Maachi M, Lagathu C, Kim MJ, Caron M, Vidal H, Capeau J, Feve B. Recent advances in the relationship between obesity, inflammation, and insulin resistance. Eur Cytokine Netw 17: 4–12, 2006. - PubMed
-
- Boden G Role of fatty acids in the pathogenesis of insulin resistance and NIDDM. Diabetes 46: 3–10, 1997. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Miscellaneous