Targeted disruption of ephrin B1 in cells of myeloid lineage increases osteoclast differentiation and bone resorption in mice - PubMed
Targeted disruption of ephrin B1 in cells of myeloid lineage increases osteoclast differentiation and bone resorption in mice
Shaohong Cheng et al. PLoS One. 2012.
Abstract
Disruption of ephrin B1 in collagen I producing cells in mice results in severe skull defects and reduced bone formation. Because ephrin B1 is also expressed during osteoclast differentiation and because little is known on the role of ephrin B1 reverse signaling in bone resorption, we examined the bone phenotypes in ephrin B1 conditional knockout mice, and studied the function of ephrin B1 reverse signaling on osteoclast differentiation and resorptive activity. Targeted deletion of ephrin B1 gene in myeloid lineage cells resulted in reduced trabecular bone volume, trabecular number and trabecular thickness caused by increased TRAP positive osteoclasts and bone resorption. Histomorphometric analyses found bone formation parameters were not changed in ephrin B1 knockout mice. Treatment of wild-type precursors with clustered soluble EphB2-Fc inhibited RANKL induced formation of multinucleated osteoclasts, and bone resorption pits. The same treatment of ephrin B1 deficient precursors had little effect on osteoclast differentiation and pit formation. Similarly, activation of ephrin B1 reverse signaling by EphB2-Fc treatment led to inhibition of TRAP, cathepsin K and NFATc1 mRNA expression in osteoclasts derived from wild-type mice but not conditional knockout mice. Immunoprecipitation with NHERF1 antibody revealed ephrin B1 interacted with NHERF1 in differentiated osteoclasts. Treatment of osteoclasts with exogenous EphB2-Fc resulted in reduced phosphorylation of ezrin/radixin/moesin. We conclude that myeloid lineage produced ephrin B1 is a negative regulator of bone resorption in vivo, and that activation of ephrin B1 reverse signaling inhibits osteoclast differentiation in vitro in part via a mechanism that involves inhibition of NFATc1 expression and modulation of phosphorylation status of ezrin/radixin/moesin.
Conflict of interest statement
Competing Interests: The authors have declared that no competing interests exist.
Figures
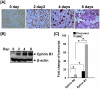
[A–B]: Expression of ephrin B1 is increased during osteoclast differentiation. Osteoclast precursors isolated from non-adherent bone marrow of wild-type (WT) mice were treated with 20 ng/ml of M-CSF and 30 ng/ml of RANKL for 0, 2, 4 and 8 days. The differentiated multinuclear cells (MNCs) were monitored by TRAP staining. The parallel cultures were harvested, and total cellular proteins were extracted for Western blot. [C]: Expression of ephrin B1 is more abundant than ephrin B2 in osteoclasts. Mouse splenocytes were treated with M-CSF only or M-CSF plus RANKL for 4 days. Total RNA was extracted for real-time PCR. Values are fold-change over the expression level of ephrin B2 transcript in undifferentiated precursors, and expressed as mean ± SEM (standard error of mean) (n = 3). A star represents statistical significance of expression level of transcript in MNCs as compared to the precursors or between two groups indicated (P<0.01).
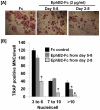
[A]: Osteoclast precursors derived from WT mice were differentiated in the presence of 20 ng/ml of M-CSF and 30 ng/ml of RANKL for 8 days. The cells were also treated with clustered EphB2-Fc during day 2–8 or day 5–8 of the culture as indicated in the figure, followed by TRAP staining. [B]: Quantitative data of TRAP positive MNCs. Values are mean ± SEM (n = 8). A star presents statistical significance of MNCs in the differentiated cultures treated with EphB2-Fc as compared to the cultures treated with control Fc (P<0.01).
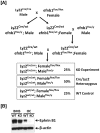
[A]: Schematic diagram of generation of ephrin B1 conditional KO and control WT. Mice with ephrin B1 deletion in myeloid lineage cells are generated by crossing ephrin B1 loxp mice with Lyz2-Cre knock-in mice. [B]: Ephrin B1 is not expressed in osteoclasts from the myeloid-specific conditional KO mice. Splenocytes derived from WT and KO mice were cultured in the presence of 20 ng/ml of M-CSF and 30 ng/ml of RANKL for 3 days. Bone marrow stromal (BMS) cells isolated from the long bones were also cultured in α-MEM medium containing 10% FBS for 6 days. Osteoclasts (OC) and BMS cells were harvested, respectively, and the cellular proteins were extracted for measurement of ephrin B1 protein, by Western blot.
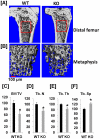
[A]: Longitudinal section of μ-CT images of distal femurs of WT and KO mice. The metaphysis of distal femurs were selected for analyses of trabecular bone parameters by μ-CT. [B]: μ-CT images of trabecular bone of the distal metaphysis of the femurs. [C–F]: Quantitative measurements of trabecular bone at the metaphysis of distal femurs. [C]: Percentage change of trabecular bone volume/total bone volume (BV/TV) of the distal femur of KO mice as compared to WT littermate controls. [D]: Percentage change of trabecular number (Tb. N) of the distal femur of KO mice as compared to WT littermate controls. [E]: Percentage change of trabecular thickness (Tb. Th) of the distal femur of KO mice as compared to WT littermate controls. [F]: Percentage change of trabecular separation (Tb. Sp) of the distal femur of KO mice as compared to WT littermate controls. Values are expressed as mean ± SEM (n = 8). A star presents statistical significance (P<0.05) as compared to WT littermate controls.
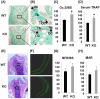
[A–B]: TRAP staining of osteoclasts at trabecular surface of metaphysis of the femur in WT and KO mice at 21 weeks of age. Bone section was counterstained with methyl green. Arrows denote representative TRAP positive osteoclasts. A: 20× magnification, B: 100× magnification. [C]: Percentage change of TRAP labeled surface/trabecular bone surface (Oc. S/BS) in the bone of ephrin B1 KO mice as compared to WT controls at 21 weeks of age. Values are presented as mean ± SEM (n = 8). [D]: Percentage change of serum TRAP activity of ephrin B1 KO mice as compared to WT controls at 21 weeks of age. Values are presented as mean ± SEM (n = 8). [E–F]: Trabecular bone formation is unaffected in mice with deletion of ephrin B1 in myeloid lineage cells. E: Images of H & E staining of distal femurs of WT and KO mice (20×). F: representative images of calcein double labeled trabecular bone of WT and KO mice (200×). [G–H]: Quantitative data of trabecular bone formation measured at distal femur metaphysis of WT and KO mice at 21 weeks of age. G: Bone formation rate/bone surface (BFR/BS). Values are presented as mean ± SEM (n = 8). H: Mineral apposition rate (MAR). Values are presented as mean ± SEM (n = 8).
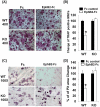
[A]: Interaction of ephrin B1 with EphB2-Fc suppresses osteoclast differentiation. Splenocytes from WT and ephrin B1 KO mice were cultured in the presence of RANKL and M-CSF for 24 hours, and then treated with soluble EphB2-Fc (2 µg/ml) or control Fc for 8 days. TRAP staining was performed after 6 days EphB2-Fc treatment. MNCs were visualized under microscope. [B]: Quantification of MNCs (3 or more nuclei per cell). Values are mean ± SEM (n = 8). A star presents statistical significance (P<0.01) as compared to the cells treated with control Fc. [C]: Interaction of ephrin B1 with EphB2-Fc suppresses bone resorption. Splenocytes from WT and ephrin B1 KO mice were seeded on bone slices, and cultured in the presence of RANKL and M-CSF for 24 hours, and then treated with EphB2-Fc or control Fc for additional 8 days. Resorption pits on bone slices were analyzed after 8 days of EphB2-Fc treatment. [D]: Quantification of resorption pit areas. Values are mean ± SEM (n = 6). A star presents statistical significance (P<0.01) as compared to the cells treated with control Fc.
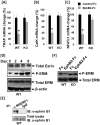
[A–C]: Treatment of EphB2-Fc inhibits expression of osteoclast differentiation marker genes. Precursors were cultured in the presence of RANKL and M-CSF for 24 hours, and then treated with soluble EphB2-Fc (2 µg/ml) or control Fc for another 4 days. Total RNA was extracted for real-time RT-PCR using specific primers to TRAP, cathepsin D (CatK) and NFATc1. Values are expressed as fold change over WT cells ± SEM (n = 3). A star presents statistical significance (P<0.05) as compared to the cells derived from WT littermate mice. [D]: ERM phosphorylation is increased during RANKL induced osteoclast differentiation. Splenocytes from WT mice were treated with 20 ng/ml of M-CSF and 30 ng/ml of RANKL for 0, 2, 4 and 8 days. Total cellular proteins from osteoclasts were extracted for Western blot. [E]: Ephrin B1 interacts with NHERF1 in osteoclasts. Splenocytes were treated with M-CSF and RANKL for 4 days. Cells were then treated with EphB2-Fc for 5 minutes, and used for immunoprecipitation. [F]: Activation of ephrin B1 reverse signaling inhibits ERM phosphorylation. Splenocytes derived from ephrin B1 KO and WT mice were treated with M-CSF and RANKL for 4 days, and then the cells were treated with 2 µg/ml of EphB2-Fc or Fc in differentiation medium for another 24 hours. Total cellular proteins were extracted for Western blot.
Similar articles
-
Xing W, Kim J, Wergedal J, Chen ST, Mohan S. Xing W, et al. Mol Cell Biol. 2010 Feb;30(3):711-21. doi: 10.1128/MCB.00610-09. Epub 2009 Dec 7. Mol Cell Biol. 2010. PMID: 19995908 Free PMC article.
-
Cheng S, Kesavan C, Mohan S, Qin X, Alarcon CM, Wergedal J, Xing W. Cheng S, et al. PLoS One. 2013 Jul 11;8(7):e69051. doi: 10.1371/journal.pone.0069051. Print 2013. PLoS One. 2013. PMID: 23874863 Free PMC article.
-
Lu J, Shi X, Fu Q, Han Y, Zhu L, Zhou Z, Li Y, Lu N. Lu J, et al. Cell Mol Biol Lett. 2024 Jul 8;29(1):100. doi: 10.1186/s11658-024-00614-5. Cell Mol Biol Lett. 2024. PMID: 38977961 Free PMC article.
-
Osteoclast differentiation by RANKL and OPG signaling pathways.
Udagawa N, Koide M, Nakamura M, Nakamichi Y, Yamashita T, Uehara S, Kobayashi Y, Furuya Y, Yasuda H, Fukuda C, Tsuda E. Udagawa N, et al. J Bone Miner Metab. 2021 Jan;39(1):19-26. doi: 10.1007/s00774-020-01162-6. Epub 2020 Oct 20. J Bone Miner Metab. 2021. PMID: 33079279 Review.
-
Roles of Wnt signals in bone resorption during physiological and pathological states.
Maeda K, Takahashi N, Kobayashi Y. Maeda K, et al. J Mol Med (Berl). 2013 Jan;91(1):15-23. doi: 10.1007/s00109-012-0974-0. Epub 2012 Oct 31. J Mol Med (Berl). 2013. PMID: 23111637 Review.
Cited by
-
Cheng S, Aghajanian P, Pourteymoor S, Alarcon C, Mohan S. Cheng S, et al. Sci Rep. 2016 Oct 24;6:35748. doi: 10.1038/srep35748. Sci Rep. 2016. PMID: 27775044 Free PMC article.
-
Regulation of cell differentiation by Eph receptor and ephrin signaling.
Wilkinson DG. Wilkinson DG. Cell Adh Migr. 2014;8(4):339-48. doi: 10.4161/19336918.2014.970007. Cell Adh Migr. 2014. PMID: 25482623 Free PMC article. Review.
-
Cheng S, Xing W, Pourteymoor S, Schulte J, Mohan S. Cheng S, et al. Endocrinology. 2016 Jan;157(1):127-40. doi: 10.1210/en.2015-1473. Epub 2015 Nov 12. Endocrinology. 2016. PMID: 26562260 Free PMC article.
-
Popov C, Kohler J, Docheva D. Popov C, et al. Front Aging Neurosci. 2016 Jan 6;7:246. doi: 10.3389/fnagi.2015.00246. eCollection 2015. Front Aging Neurosci. 2016. PMID: 26779014 Free PMC article.
-
Mechanisms of ephrin-Eph signalling in development, physiology and disease.
Kania A, Klein R. Kania A, et al. Nat Rev Mol Cell Biol. 2016 Apr;17(4):240-56. doi: 10.1038/nrm.2015.16. Epub 2016 Jan 21. Nat Rev Mol Cell Biol. 2016. PMID: 26790531 Review.
References
-
- Jensen PL. Eph receptors and ephrins. Stem Cells. 2000;18:63–64. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous