The continued threat of emerging flaviviruses - PubMed
Review
The continued threat of emerging flaviviruses
Theodore C Pierson et al. Nat Microbiol. 2020 Jun.
Abstract
Flaviviruses are vector-borne RNA viruses that can emerge unexpectedly in human populations and cause a spectrum of potentially severe diseases including hepatitis, vascular shock syndrome, encephalitis, acute flaccid paralysis, congenital abnormalities and fetal death. This epidemiological pattern has occurred numerous times during the last 70 years, including epidemics of dengue virus and West Nile virus, and the most recent explosive epidemic of Zika virus in the Americas. Flaviviruses are now globally distributed and infect up to 400 million people annually. Of significant concern, outbreaks of other less well-characterized flaviviruses have been reported in humans and animals in different regions of the world. The potential for these viruses to sustain epidemic transmission among humans is poorly understood. In this Review, we discuss the basic biology of flaviviruses, their infectious cycles, the diseases they cause and underlying host immune responses to infection. We describe flaviviruses that represent an established ongoing threat to global health and those that have recently emerged in new populations to cause significant disease. We also provide examples of lesser-known flaviviruses that circulate in restricted areas of the world but have the potential to emerge more broadly in human populations. Finally, we discuss how an understanding of the epidemiology, biology, structure and immunity of flaviviruses can inform the rapid development of countermeasures to treat or prevent human infections as they emerge.
Figures

a. Flaviviruses encode a single open reading frame that is translated at the endoplasmic reticulum into a polyprotein, which subsequently is cleaved by viral and host cell proteases. This processing results in ten functional proteins including the three structural proteins C, prM, and E and seven non-structural proteins. NS4A exists in two forms that differ with respect to cleavage of the 2K domain at its carboxy-terminus. b. Flavivirus E proteins are elongated three domain structures tethered to the viral membrane by a stem and two antiparallel transmembrane domains. E protein domains are indicated in red, yellow, and blue (domain I-III, respectively). The M protein, also attached to the viral membrane by two transmembrane domains, is shown in purple. c. The distinct arrangement of E proteins on immature (left) and mature (right) forms of the virion are depicted.
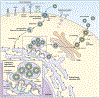
Flaviviruses infect mammalian cells via interactions with multiple types of host attachment factors, including molecules that bind to the viral membrane or virion-associated N-linked carbohydrates. Interactions with cell surface host factors, such as CLEC5a, may also initiate signaling pathways that modulate the host immune response. Virions are internalized by clathrin-dependent mechanisms that usurp host factors involved in the uptake of large macromolecules, including RNASEK. Viral fusion with host membranes occurs in the endosome in a low pH-dependent manner. Viral RNA replication occurs on membranes of the host reorganized through the actions of the non-structural proteins. These virus-induced membrane structures spatially coordinate viral genomic RNA replication and virion morphogenesis, and shield replication products from host innate immune sensors. Virus particles assemble at and bud into the endoplasmic reticulum and traffic out of the cell. Virion maturation, defined by the cleavage of prM by a furin-like protease, occurs during egress.

Flaviviruses cause different febrile syndromes depending on the virus and the affected patient. Several flaviviruses are neurotropic (e.g., West Nile, Japanese encephalitis, TBEV, Usutu, Zika, and Ilheus), can spread to the brain and spinal cord, and cause severe neurological syndromes including meningitis, encephalitis, and acute flaccid paralysis. These can result in death or long-term disability in survivors. Other flaviviruses (e.g., Yellow fever, Dengue, and Zika) cause visceral disease resulting in liver failure, hemorrhagic syndromes, and vascular compromise, also resulting in death. Uniquely, ZIKV can infect the tissues of the male and female reproductive tracts leading to sexual transmission. ZIKV infection during pregnancy can cause injury to the placenta as well as transmit to the developing fetus, resulting in placental insufficiency, microcephaly, congenital malformations, and fetal demise.
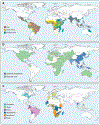
a. The global distribution of Aedes-transmitted flaviviruses ZIKV, YFV, and DENV are shown. b. The global distribution JEV and WNV is shown c. The approximate geographic locations of flaviviruses with the potential for emergence in human populations.
Similar articles
-
Nikolay B. Nikolay B. Trans R Soc Trop Med Hyg. 2015 Oct;109(10):609-18. doi: 10.1093/trstmh/trv066. Epub 2015 Aug 17. Trans R Soc Trop Med Hyg. 2015. PMID: 26286946 Review.
-
INFECTIOUS DISEASES. An obscure mosquito-borne disease goes global.
Enserink M. Enserink M. Science. 2015 Nov 27;350(6264):1012-3. doi: 10.1126/science.350.6264.1012. Science. 2015. PMID: 26612926 No abstract available.
-
Anticipating Emerging Mosquito-borne Flaviviruses in the USA: What Comes after Zika?
Evans MV, Murdock CC, Drake JM. Evans MV, et al. Trends Parasitol. 2018 Jul;34(7):544-547. doi: 10.1016/j.pt.2018.02.010. Epub 2018 Mar 15. Trends Parasitol. 2018. PMID: 29551358 Free PMC article.
-
Mackenzie JS, Williams DT. Mackenzie JS, et al. Zoonoses Public Health. 2009 Aug;56(6-7):338-56. doi: 10.1111/j.1863-2378.2008.01208.x. Zoonoses Public Health. 2009. PMID: 19486319 Review.
-
Weissenböck H, Hubálek Z, Bakonyi T, Nowotny N. Weissenböck H, et al. Vet Microbiol. 2010 Jan 27;140(3-4):271-80. doi: 10.1016/j.vetmic.2009.08.025. Epub 2009 Aug 26. Vet Microbiol. 2010. PMID: 19762169 Review.
Cited by
-
Deep mining reveals the diversity of endogenous viral elements in vertebrate genomes.
Nino Barreat JG, Katzourakis A. Nino Barreat JG, et al. Nat Microbiol. 2024 Nov;9(11):3013-3024. doi: 10.1038/s41564-024-01825-4. Epub 2024 Oct 22. Nat Microbiol. 2024. PMID: 39438719 Free PMC article.
-
Hassert M, Steffen TL, Scroggins S, Coleman AK, Shacham E, Brien JD, Pinto AK. Hassert M, et al. J Virol. 2021 Jul 26;95(16):e0057321. doi: 10.1128/JVI.00573-21. Epub 2021 Jul 26. J Virol. 2021. PMID: 34076486 Free PMC article.
-
Structure-guided paradigm shifts in flavivirus assembly and maturation mechanisms.
Nicholls CMR, Sevvana M, Kuhn RJ. Nicholls CMR, et al. Adv Virus Res. 2020;108:33-83. doi: 10.1016/bs.aivir.2020.08.003. Epub 2020 Sep 23. Adv Virus Res. 2020. PMID: 33837721 Free PMC article. Review.
-
Balancing functions of regulatory T cells in mosquito-borne viral infections.
Sann S, Kleinewietfeld M, Cantaert T. Sann S, et al. Emerg Microbes Infect. 2024 Dec;13(1):2304061. doi: 10.1080/22221751.2024.2304061. Epub 2024 Jan 25. Emerg Microbes Infect. 2024. PMID: 38192073 Free PMC article. Review.
-
Machmouchi D, Courageot MP, Ogire E, Redecke L, Kohl A, Desprès P, Roche M. Machmouchi D, et al. PLoS Negl Trop Dis. 2024 Aug 23;18(8):e0012146. doi: 10.1371/journal.pntd.0012146. eCollection 2024 Aug. PLoS Negl Trop Dis. 2024. PMID: 39178324 Free PMC article.
References
-
- Gubler DJ Dengue/dengue haemorrhagic fever: history and current status. Novartis Found Symp 277, 3–16; discussion 16-22, 71-13, 251-253 (2006). - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources